Research, case reports, clinical trials
» DEUTSCHAntabuse (disulfiram) against Cancer
a presentation by Boris Cvek, Ph.D.
University of Olomouc, Social Health Institute OUSHI
(formerly at the Department of Cell Biology and Genetics)
Research in cells and animals
Many scientific publications have demonstrated that Antabuse (disulfiram), especially when combined with copper (cf. The Active Compound further below), kill cancer cells and is able to suppress tumors in mice.
The figure below, from our research now being prepared for publication (NOTE, Dec’17 – in between published: Nature, Dec 6/14 2017, doi:10.1038/nature25016, see Alcohol-abuse drug Antabuse (disulfiram) kills cancer cells), shows mice with a metastasis of human breast cancer, most effectively suppressed by the active compound.

gluCu = food supplement containing copper | Mock = control group
Case report by Dr. Lewison (Prog. Clin. Biol. Res. 12, 47-53, 1977)
Spontaneous regression of breast cancer, Prog Clin Biol Res 1977, Lewison EF
In 1977, Dr. Lewison from Johns Hopkins University in the USA published a case report of a patient with metastasizing breast cancer who became an alcoholic. Between 1961-1971 she used Antabuse (Disulfiram) and became cancer-free. She died in 1971 – not because of cancer, but by falling from a window while heavily drunk.
“However, in 1961 she became a severe alcoholic and it was necessary to discontinue all hormone therapy and Antabuse was started [for treatment of alcoholism]. Over the next 10 years, from 1961 to 1971, complete resolution of all bone lesions in the spine, skull, pelvis and ribs gradually occurred and the patient remained clinically free of cancer with no further hormone therapy, chemotherapy, or radiation therapy. Frequent psychiatric care was required and she remained on and off Antabuse therapy for her continued drinking problem. She died in 1971 when she accidentally fell from a third floor window. The coroner’s report showed a high blood alcohol level and residual nests of metastatic carcinoma in the bone marrow.”
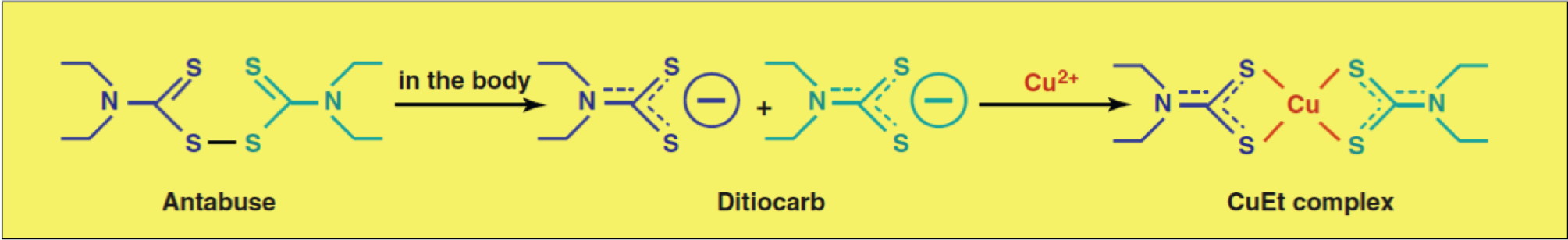
Ditiocarb clinical trial (Biotherapy 6, 9-13, 1993)
Sodium dithiocarb as adjuvant immunotherapy for high risk breast cancer: a randomized study, Biotherapy 1993 6(1) 9-12
In 1993, French scientists published a phase II clinical trial of a drug called Ditiocarb. This compound is produced after ingestion of Antabuse. They used very low doses – 700 mg weekly, in comparison to a standard Antabuse dosage of 250-500 mg daily.
Sixty-four women with non-metastatic high risk breast cancer after surgery were divided into two equal groups. The first group were placed on standard chemo + placebo, while the second were on the same chemo + Ditiocarb. The drugs were taken for 9 months. After 5 years, 55% of patients were alive in the first group compared to 81% of patients in the second group.
It is to be expected that with higher doses, and after longer treatment, the effect might be much stronger. Our hypothesis is that the active compound (which occurs in the body after Ditiocarb reaction with copper, cf. The Active Compound below) was able to destroy micro metastases in some patients – even at such low doses and after only 9 months.
Case report from Utah (Mol. Cancer Ther. 3, 1049-1060, 2004)
In 2004, scientists from Utah published a case report of a patient with melanoma metastasis in the liver, which – by using a combination of disulfiram and zinc – shrank after a few months and disappeared after three and a half years.

Today, the fact that the active compound is a copper combination is already known (in fact, the zinc compound does not exist in human body and disulfiram prefers copper that is naturally occurring in the body).
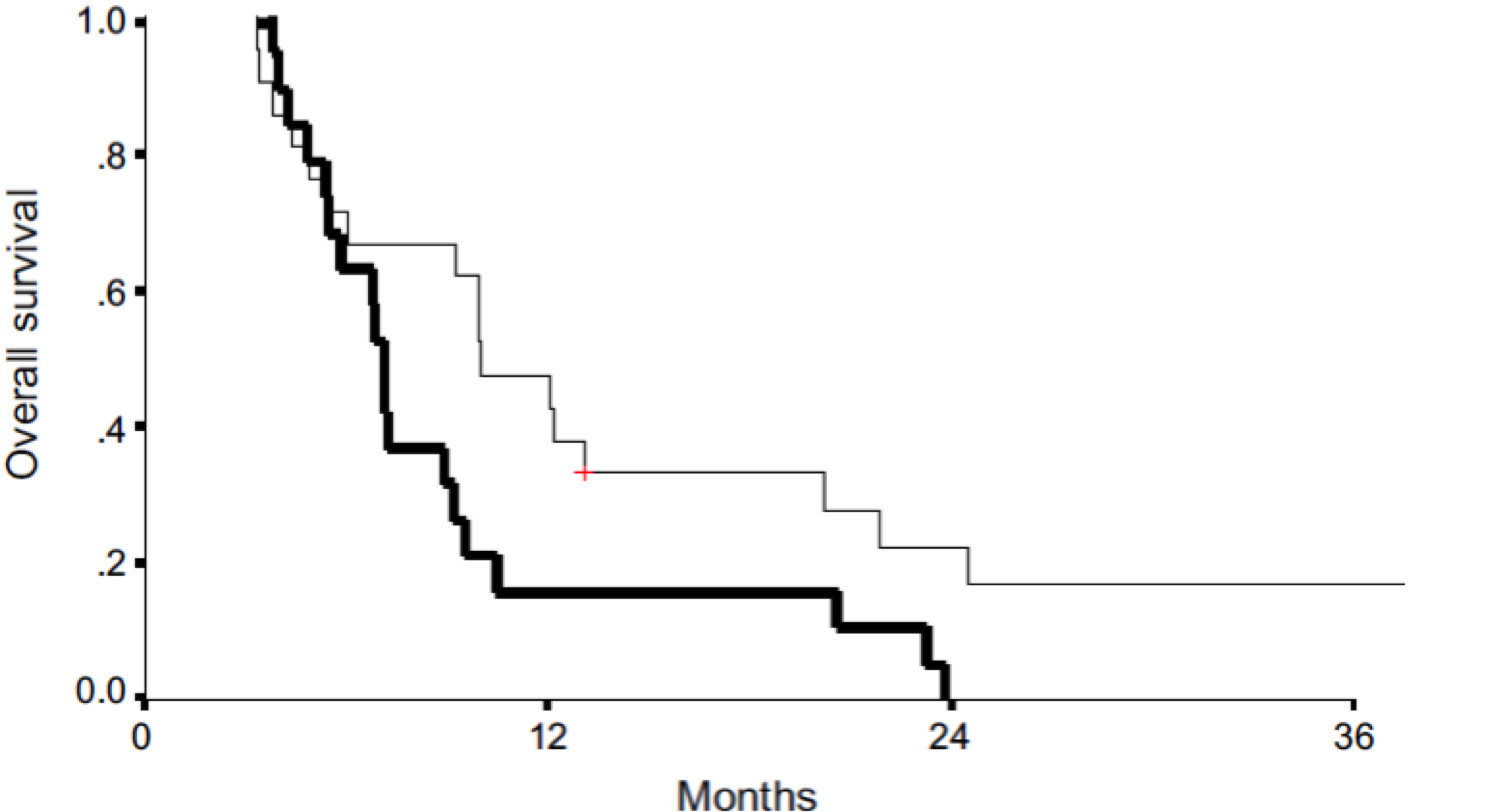
Disulfiram clinical trial (Oncologist 20, 366-367, 2015)
A phase IIb trial assessing the addition of disulfiram to chemotherapy for the treatment of metastatic non-small cell lung cancer, Oncologist 2015 Apr 20(4) 366-7 Epub 2015 Mar 16
In 2015, the Oncologist journal published results of a clinical trial from Israel, where 40 patients with metastatic lung cancer were divided into two equal groups (of 20 each). The first group were placed on standard chemo, while the second group were on standard chemo + disulfiram (just 120 mg daily). Patients from the second group survived 3 months longer on average and, most importantly, two survived for a long time. The figure further shows that all the patients from the first group died after two years.
The active compound
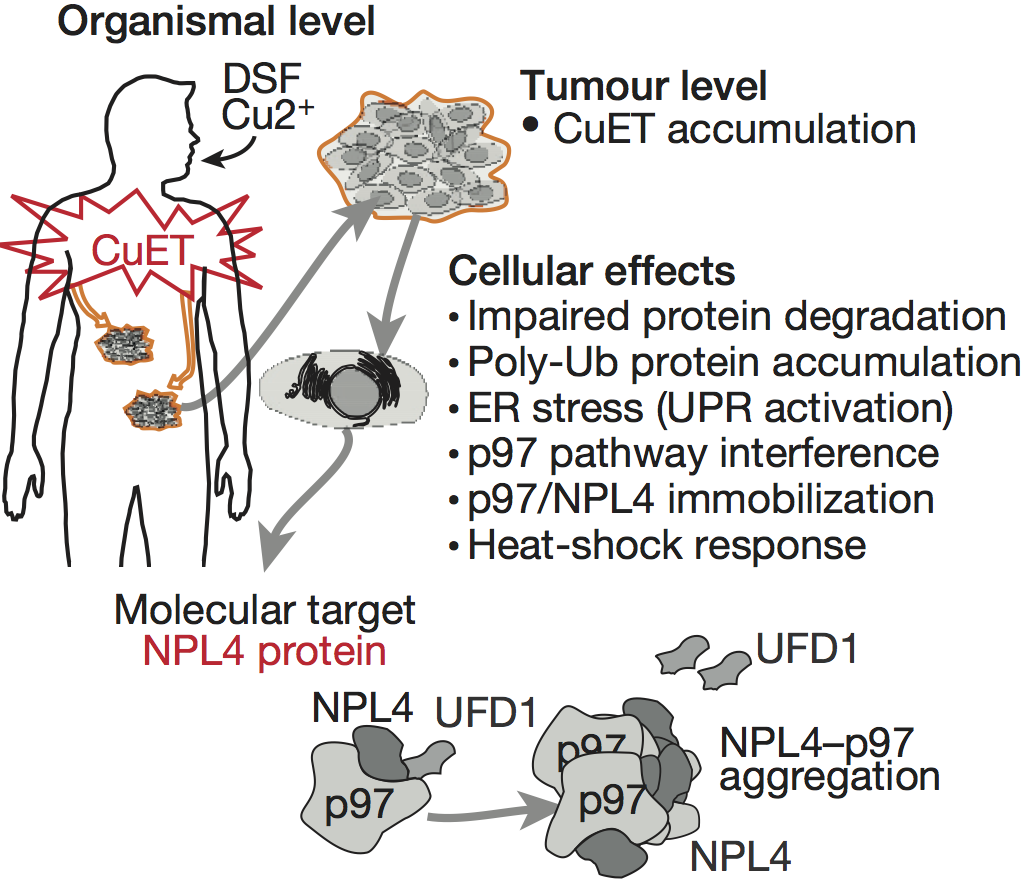
We have various experimental evidence, now prepared for publication, showing that the active compound is a complex of Ditiocarb and copper; which occurs under normal conditions after Antabuse ingestion in the human body.
Phase II and phase III clinical trials
To get approval from state authorities for Antabuse to be used normally in oncology, we need to conduct further and larger clinical trials – i.e. phase II clinical trials (hundreds of patients) and phase III in particular (thousands of patients). We need such trials to examine various cancers and to combine Antabuse with various standard chemo drugs.
Normally, clinical trials are funded by pharmaceutical companies. However, Antabuse is not patentable. The advantage is that it is affordable (hundreds of Dollars/Euros per patient per year), yet the weakness is that it is not interesting for business. Therefore, Antabuse clinical trials must be paid by charities and governments in the public interest. We want to finance such clinical trials.
If large phase III clinical trials show that Antabuse is able to cure some cancers, it would be, as an inexpensive drug, affordable even for patients in low-income countries.
First steps and further progress – including fast-track approval
In Olomouc, where we have experience with Antabuse research, there are oncologists who are willing to conduct Antabuse clinical trials. In collaboration with them, we have designed a phase II clinical trial of 100 patients with metastatic breast cancer and 100 patients with metastatic lung cancer (500 mg Antabuse + 2 mg copper daily).
In all patients, overall survival will be determined, and if their tumors shrink, do not grow, or grow further. The results will be of value for the identification of patients who are most sensitive to Antabuse therapy, and for further research as to why some patients are more sensitive than others.
We would also like to conduct further trials in patients with different cancers. In the case of positive results, we would design phase III clinical trials and talk to the FDA and EMA to win a fast-track approval of Antabuse for metastatic cancer where it is curable. Negative results would instigate phase II clinical trials of Antabuse in combination with standard chemo.
Why test Antabuse in Olomouc?
In Olomouc we have a team of oncologists led by prof. Bohuslav Melichar, head of the Oncology Unit of the Teaching Hospital, ready to conduct clinical trials of Antabuse in patients with metastatic breast/lung cancer and other cancers – as soon as sufficient funding is found.
Preliminary/rough cost estimate of the 100 + 100 trial
(subject to review / amendments)
- Wages of physicians (2 physicians), coordinator and supervisor of clinical trial
some € 80,000 per year – for 2 years € 160,000
- Material (Antabuse, copper supplementation, materials used for standard health care in hospitals), other services and non-material costs (examination of patients, insurance according to law),
some € 120,000 per year – for 2 years € 240,000
- Overall cost of 1 phase II clinical trial of 100 + 100 patients
some € 200,000 per year – for 2 years € 400,000
This project is the result of our long-term scientific research of Antabuse anti-cancer activity here in Olomouc. So far, there is only one team, besides us, focused on Antabuse application in oncology, (prof. Weiguang Wang in UK), but they are working on some patentable formulation of disulfiram.
Our strategy fits perfectly into the mission of Olomouc University Social Health Institute (OUSHI). It is not “only” about curing patients, but we have an ambition to contribute to changing healthcare systems globally, to make them more affordable with “nonprofit drugs”. More about this idea can be found in the article “Nonprofit drugs as the salvation for world‘s health care systems: the case of Antabuse” by B. Cvek, published in Drug Discovery Today.
CONTACT – Boris Cvek, Ph.D. / Prof. Peter Tavel, Ph.D. (UP / Olomouc University (CZ)
PROPOSED/POSSIBLE PARTNERS* – Prof. Ray Deshaies (California Institute of Technology, Pasadena CA (USA) / Prof. Vikas Sukhatme (Dana-Farber/Harvard Cancer Research, Boston MA (USA) / Prof. Jiří Bártek (UP, Olomouc (CZ), Danish Cancer Society Research Center, Copenhagen (DK), Karolinska Institutet, Stockholm (S) / Prof. Ping Dou (Barbara Ann Karmanos Cancer Institute, Detroit MI (USA) / Prof. Christoph Driessen (Kantonsspital St. Gallen (CH), SAKK (CH) / Prof. Pavla Poučková (1st Medical Faculty Charles University, Prague (CZ)
*chronologically, as we have “found” them and with whom we currently (2015/16) work together on our Nature publication



 During the last years we confirmed that disulfiram, also known as antabuse, is effective against cancer at cell level and in animals.
During the last years we confirmed that disulfiram, also known as antabuse, is effective against cancer at cell level and in animals.


 Jocelyn Kaiser
Jocelyn Kaiser


